
The abandoned Tulsequah Chief Mine in northwestern British Columbia has leaked toxic, acidic, metal-laden water into the Tulsequah and Taku rivers since 1957. The Tulsequah Chief Mine which produced gold, copper, lead, and zinc between 1951 and 1957, was abandoned without reclamation. Sulfide minerals present in the Tulsequah Chief deposit are pyrite, chalcopyrite, sphalerite, and galena. (Picture is USGS, 2018, Public Domain. Mineralogy of Tulsequah Chief deposit is from British Columbia Minfile Record Summary, 104K 002))
In my last posting I described how the only operating sulfide mineral mine in the northern Midwest, the Eagle Mine, has provided jobs, tax revenues, and economic activity for the Marquette, Michigan area. What is not to like about this picture?
As it turns out, there are some negative possible outcomes from sulfide mineral mining – pollution, disruption of wilderness areas, and desecration of locations Native Americans hold scared.
In this blog I enumerate environmental risks that can come from sulfide minerals. I also note that starting in the last half of the twentieth century new state and federal environmental regulations were enacted in the United States. In Michigan, Wisconsin, and Minnesota specific mining regulations now exist for sulfide mineral mining.
Future blogs will discuss the opposition of advocates of wilderness areas and of Native American to sulfide mineral mining.
Chemical reactions of sulfide minerals
When a sulfide mineral encounters oxygen, the mineral is destabilized and oxidizes. This means that the bond between the sulfur and the metal is broken, and the metal can go into an aqueous solution. The reaction also releases hydrogen ions and makes the water acidic. This presence of acid promotes the release of metals from rock. This process is natural and occurs all the time in nature; however, mining brings a large quantity of sulfur-bearing rocks to the surface and these rocks are crushed, which means that a large area of mineral surface is exposed to oxygen. This increases exponentially the oxidation process. In many instances in the nineteenth and twentieth centuries, the extraction of sulfide minerals and processing of sulfide mineral ore did not keep sulfide minerals from encountering oxygen and water.
Sulfide minerals present at various locations in the Northern Midwest are listed in a note at the end of this blog.
Acid rock and acid mine drainage
Acid and metal-rich water originating from the oxidation of sulfide minerals is called acid rock drainage (ARD). An acid drainage generated from the oxidation of sulfide minerals associated with the mining process is called acid mine drainage (AMD). The process of ARD and AMD is, however, the same.
A net simplified characterization for the reaction of sulfide minerals, using the mineral pyrite (iron sulfide), shows that when pyrite is exposed to water and oxygen, the result of this reaction is the formation of iron (ferric) hydroxide, which precipitates as an ugly yellow / orange mess called “yellow boy” or ocher. When sulfide minerals that are composed of metals other than iron are exposed to oxygen and water, the products of the reaction are sulfuric acid as well as the release of these other metals (e.g., cobalt, copper, iron, nickel, zinc, lead, etc.) that were originally bound in the sulfide mineral. Recent experimental investigations show that some metals in low concentrations – including nickel, copper, and zinc – are vital for humans, but in larger concentrations, they can be hazardous to both human health and the environment.

Although pyrite (iron sulfide) is usually used to illustrate the damage that can occur when sulfide minerals come in contact with air and water, the reaction of different sulfide minerals varies. Based on laboratory results, the following sulfide minerals can be classified in terms of their oxidation reaction based on their sulfur release rates from the highest to the lowest: gersdorffite > pyrrhotite > arsenopyrite > pyrite > fahlore > covellite > sphalerite-Fe ≥ chalcopyrite > sphalerite > galena > stibnite > chalcocite > bornite (see Chopard reference listed at the end of this blog). The implication of these laboratory results is that Michigan’s Copperwood deposit, which contains only the sulfide mineral chalcocite, should not be considered to have the same potential environmental impact as other deposits that contain more reactive sulfide minerals. The Copperwood deposit and the proposed handling of waste materials (tailings) at the proposed Copperwood Mine will be the subject of a later blog.
Consequences of changes of the chemistry of water and soils
The change in the acidity of water and soils from acid soured from sulfide minerals can be hazardous to plants, fish, and people.
Nutrients are often immobilized in the soil under acidic conditions, which hamper plant growth, microbial diversity, and their functions. AMD has also been linked to an increase in methylation. Methylation is a process in which a methyl group is added to inorganic mercury to create the most toxic form of mercury, methyl mercury. Methylation is thought to play a crucial role in the growth and development of wild rice which is a vital part of Native American culture (this concern will be the subject of a later blog).
When fish are exposed directly to metals and hydrogen ions through their gills, impaired respiration may result from chronic and acute toxicity. Fish are also exposed indirectly to metals through ingestion of contaminated sediments and food items. Yellow boy may physically coat the surface of stream sediments and stream beds destroying habitat, diminishing availability of clean gravels used for spawning, and reducing fish food items such as benthic macroinvertebrates.
The consumption of metals by fish is a potential problem for Native Americans’ diets in the Northern Midwest; fish is a staple food and fish consumption is a cultural practice that is threatened if sulfide mineral mining is allowed to add excessive amounts of metals to the environment.
The way to not encounter the problems of sulfide mineral ores is to handle them with care. Opponents of sulfide mineral mining say that this careful handling can’t be done, but new regulations have been adopted to attempt to address the problems of mining sulfide mineral ores.
Regulations for sulfide mineral mining
Pollution from sulfide mineral mines dug in the nineteenth and first half of the twentieth century are frequently used as proof that sulfide mineral mining can’t be done without harm to the environment. Criticism of sulfide mineral mining frequently fails to address modern federal and state legislation which are now in place in the United States to police proposals for mine permits and dictate mining practices once mining begins.
Starting in 1964, the following federal laws were passed in the United States: Wilderness Act of 1964, National Environmental Policy Act of 1970, Clean Air Act of 1963, Endangered Species Act of 1973, Safe Drinking Water Act of 1974, Surface Mining Control and Reclamation Act of 1977, and Toxic Substance Control Act of 1976. Consideration of one or more of these federal laws can apply to sulfide mineral mining.
In 1992, in anticipation of the mining of sulfide mineral ores, Minnesota adopted specific rules for the permitting, operating, and reclamation of sulfide mineral mining. Wisconsin passed their “prove it first law” in 1998, which blocked the issuance of any mining permits unless the applicant could provide verifiable evidence that it had operated another sulfide mineral mine for ten years without the pollution of ground or surface water. Although the “prove it first law” was repealed in 2017, Wisconsin continues to have detailed requirements for sulfide mineral mining. In 2004 Michigan adopted specific regulations for the mining of sulfide mineral ores in response to the sulfide mineral deposit that had been discovered on the Yellow Dog Plains north of Marquette (the Eagle deposit).
As of February, 2026, opponents of sulfide mineral mining have used state and federal regulation as a basis to appeal the granting of mining permits being issued for the NorthMet project in Minnesota and the Back Forty project in Michigan. The NorthMet project has now been trying to obtain a permit to mine for over twenty years. Regulations for permitting a sulfide mineral mine in Minnesota require input / or action by the Minnesota Department of Natural Resources (DNR), the United States Environmental Protection Agency ((EPA), the U.S. Army Corps of Engineers (Corps), U.S. Forest Service (USFS), and the Minnesota Pollution Control Agency (MPCA). Opponents to mining have recourse in Federal and State Courts.
Sulfide mineral mine approvals in Michigan
Despite objections of Native Americans, environmental groups (the Yellow Dog Watershed Preserve and others), and the well-funded efforts of the exclusive Huron Mountain Club, the Eagle Mine, which filed permit applications in 2006, was approved. In 2014 the Michigan Court of Appeals affirmed the Michigan Department of Environmental Quality decision to grant a mining permit for the Eagle Mine. The eight years it took for the Eagle Mine to gain a mining permit can now be expressed as “warp speed” when compared to the time it has taken other permit requests to be considered for sulfide mineral mines.
In Michigan the Copperwood Mine received its initial permits in 2012 and as of 2025, 13 years later, was still receiving environmental approvals with an updated air permit received in 2025. Although the owners of the Copperwood Mine project represent in 2026 that it has all the necessary permits to begin mining, the project still needs to receive approval for design of the facility where mining waste (tailings) will be stored.
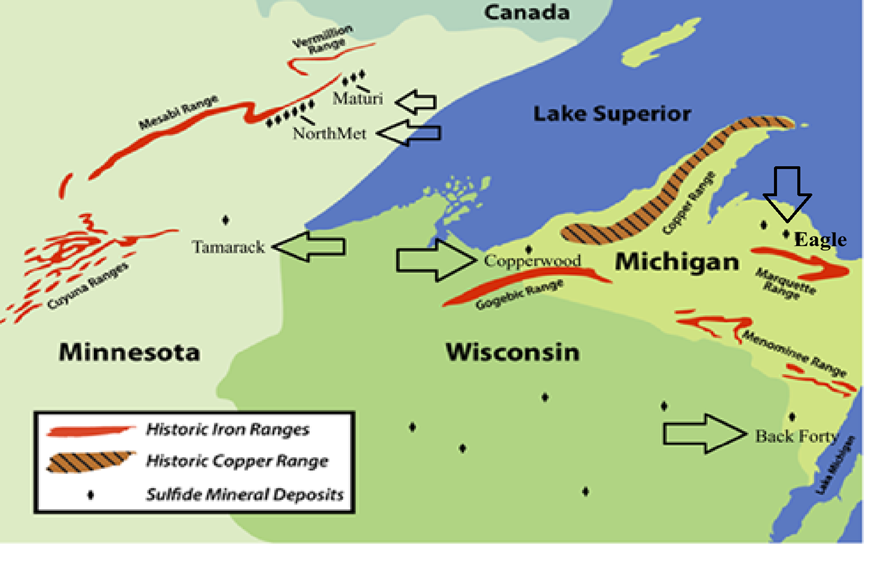
Locations of sulfide mineral deposits
Minnesota’s North Tamarack and NorthMet Projects
The Minnesota sulfide mineral deposit, the Tamarack Mine project, appears to be on a path for approval for their sulfide mineral mine. In a presentation dated December 2025 posted online, Talon Metals said they expected their Environmental Impact statement filing for the Tamarack project to be complete in 280 days (the start date for the 280 days is vague) and their facility in North Dakota for the waste (tailings) from their Minnesota Tamarack Mine to have its necessary environmental approvals in 12 to 18 months.
Given the difficulties of sulfide mineral mines from receiving approval to mine in Minnesota as demonstrated by the now 20-year purgatory of the NorthMet project, it will be interesting to watch how Talon Metals manages to navigate the sulfide mineral mine regulatory gauntlet in Minnesota. Talon Metals’ waste handling facility in North Dakota is the first of its kind, and therefore there is no precedent to use to estimate the time it will take to receive this North Dakota approval.
As of February 2026, the Minnesota Department of Natural Resources shows the regulatory process for the approval of the Tamarack Mine as only beginning with many parts of the approval process with variable (vague) time frames.

Timeline from https://files.dnr.state.mn.us/input/environmentalreview/tamarack-nickel/tal-timeline.pdf
Notes
Sulfide minerals present at the project sites in the Northern Midwest
The chemical formulas and locations of sulfide minerals present at the projects discussed in this blog are listed below. The chemical formulas use the standard chemical letter notation where Cu = copper, Fe = iron, Ni = nickel, Pb = lead, S = Sulfur, and Zn = zinc. The numbers next to the letters refer to how many atoms of an element are present in the compound. For example, bornite with the formula Cu5FeS4 has five atoms of copper, one atom of iron, and four atoms of sulfur.
| MINERAL | FORMULA | LOCATIONS | |||
| bornite | Cu5FeS4 | 1 | |||
| chalcocite | Cu2S | 2,6 | |||
| chalcopyrite | CuFeS2 | 1,3,4,5,6,7,8,9,10 | |||
| cubanite | CuFe2S3 | 4,5,8,9,10 | |||
| galena | PbS | 1,3 | |||
| millerite | NiS | 4,5 | |||
| pentlandite | (Fe,Ni)9S2 | 4,5,7,8,9,10 | |||
| pyrite | FeS2 | 1,3,6 | |||
| pyrrhotite | Fe1-xS | 4,5,8,9,10 | |||
| sphalerite | (Zn,Fe)S | 1,3,6 | |||
| talnakhite | (Cu9(Fe, Ni)8S16) | 9 | |||
| Locations | |||||
| 1 Back Forty | 6 Flambeau | ||||
| 2 Copperwood | 7 Keel Zone (Eagle Mine Area) | ||||
| 3 Crandon | 8 NorthMet | ||||
| 4 Eagle | 9 Maturi | ||||
| 5 Eagle East | 10 Tamarack | ||||
Project locations are shown on the map in this blog except for the Wisconsin Crandon deposit which is marked but not labeled.
For more information on the reactions of sulfide minerals see:
Aurelie Chopard, Mostafa Benzaazoua, Benoit Plante. Kinetic Tests to Evaluate the Relative Oxidation Rates of Various Sulfides and Sulfosalts. Conference: ICARD – IMWA 2015, 2015. https://www.researchgate.net/publication/307509806_Kinetic_Tests_to_Evaluate_the_Relative_Oxidation_Rates_of_Various_Sulfides_and_Sulfosalts.
Nordstrom, D. Kirk. “Sulfide Mineral Oxidation.” In Encyclopedia of Geobiology. Springer, 2011. https://pubs.usgs.gov/publication/70198937.
Donald Rimstidh, John A. Chermak, and Patrick M. Gagen. Rates of Reaction of Galena, Sphalerite, Chalcopyrite, and Arsenopyrite with Fe (III) in Acidic Solutions. Alpers and Blowes; Environmental Geochemistry of Sulfide Oxidation. American Chemical Society, 1994.
Next Blog
In my next blog, I review the claim that the Copperwood Mine in Michigan should not be dug because if allowed to operate it would destroy a wilderness area.



